
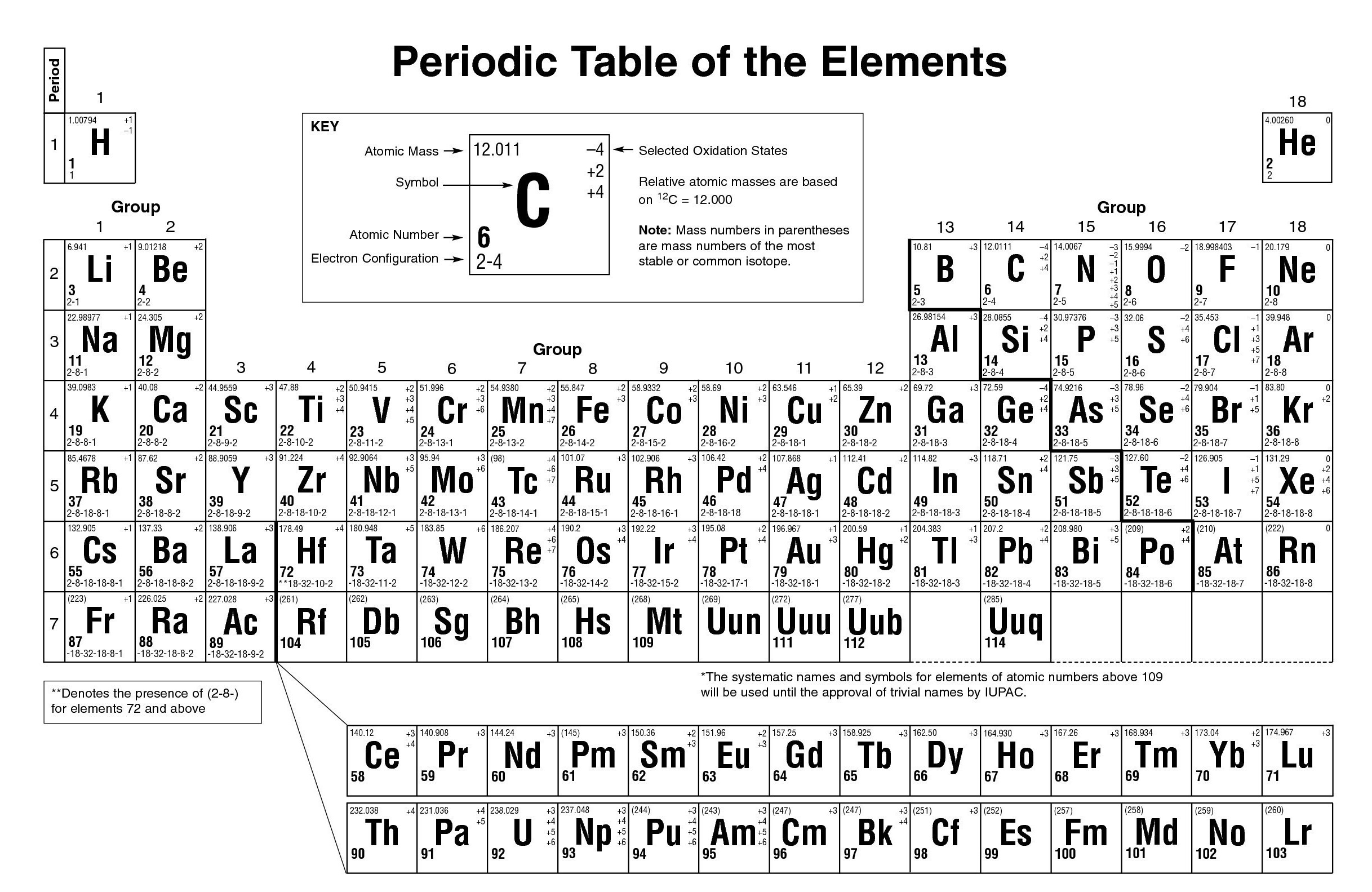
The following table gives the list of 118 elements along with their symbols and atomic number. This is why the knowledge of atomic numbers is important in understanding the chemistry of elements.
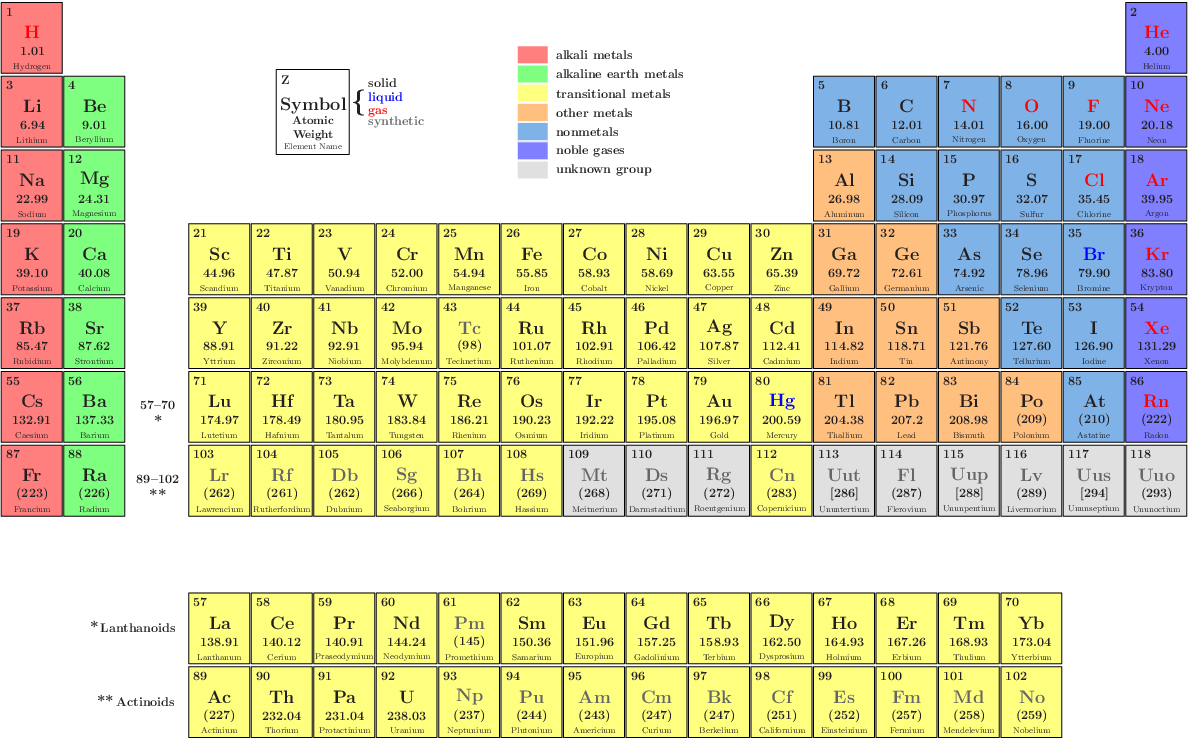
The chemical properties of an element are determined by the number of protons in the nucleus. The atomic number is the total number of protons present in the nucleus of an atom. The sum of protons and neutrons gives the atomic mass of an element. The nucleus is surrounded by negatively charged electrons. Neutrons are neutral so that it doesn’t have any charge on them. The centre of the atom is also called the nucleus which is positively charged and consists of protons and neutrons. It consists of electrons, protons and neutrons. An atom is the smallest indivisible unit of matter. An element is made up of a single type of atom. The concept of atomic number and Valency can only be understood if you know what exactly elements are made up of. It is important to know the atomic number and electronic configuration of an element to find its valency. The valency of an element is determined by the number of electrons in the valence shell. The number of electrons in the outermost shell is called valence electrons and the outermost shell is called the valence shell. This ability of an atom to gain or lose electrons to achieve a stable configuration or inert gas configuration is called the Valency of an element. of a noble gas, the atom of an element tries to gain or lose electrons. In order to achieve the most stable configuration i.e. Without the symbols, it would have been a herculean task to represent all these 118 elements and the umpteen numbers of compounds they form. For example, the element with atomic number 110 was named as ‘un un nilium’ with the symbol 'Uun', now it is named Ds.Īs far as students are concerned, it is important to study all the 118 elements with their Symbol and Valency. Chemical formulas and equations are also represented using those symbols. The elements which are new are temporarily named according to their atomic numbers. Example : 'N' represents Nitrogen, 'O' represents oxygen, etc. Example: 'Ca' representing Calcium, 'He' representing Helium, etc. When the symbol representing an element is denoted by one letter only, it is written in uppercase. Rules or Conventions followed to denote the Element using Symbol The first letter of a symbol is capitalized with the second (or third) letters being in lowercase. The symbol 'Fe' is used to denote Iron, as the Latin word for Iron is "Ferrum". Hence gold is denoted by the symbol ‘Au’. Some symbols of a few elements are derived from their Latin or Greek names. One may ask, ‘How is the symbol of an element derived?’ We can see in the table above that most of the symbols are derived from the elements’ names, by taking either the first or first two letters from the English name of the element. Some symbols have three letters, they generally represent synthesized elements newly, with some being temporarily named like that. (Image will be Uploaded Soon) Symbol of an ElementĪ symbol representing a chemical element is a 'sign' or 'notation' that generally consists of one or two letters. Example: Elements in group 1A are soft metals that react violently with water. Elements arranged vertically in columns are called ‘Groups’ and elements arranged horizontally in rows are called ‘Periods’.įurther elements are grouped as per p eriodic trends and properties. Elements are arranged in order of increasing atomic number.Įlements of the Periodic Table are denoted by a unique symbol and not its entire name, as some elements’ names can be long and complex in nature.Įlements are arranged vertically and horizontally.Key Characteristics of the Periodic Table:

The latest Periodic Table is based on Henry Moseley's modern periodic law (Henry Moseley is an English physicist). As per the periodic law, the properties of Elements are periodic functions of their atomic numbers. The Periodic Table is made up of 118 Elements. During his time only half of the elements known to us now were known, and not all of the information about elements was fully known or accurate. This periodic table was based on the atomic mass of the elements. Dimitri Mendeleev is referred to as the Father of the periodic table put forth the first form of the Periodic Table. Scientists, Professionals, Teachers, and Students of Chemistry widely use the periodic table of elements to search for chemical elements.

